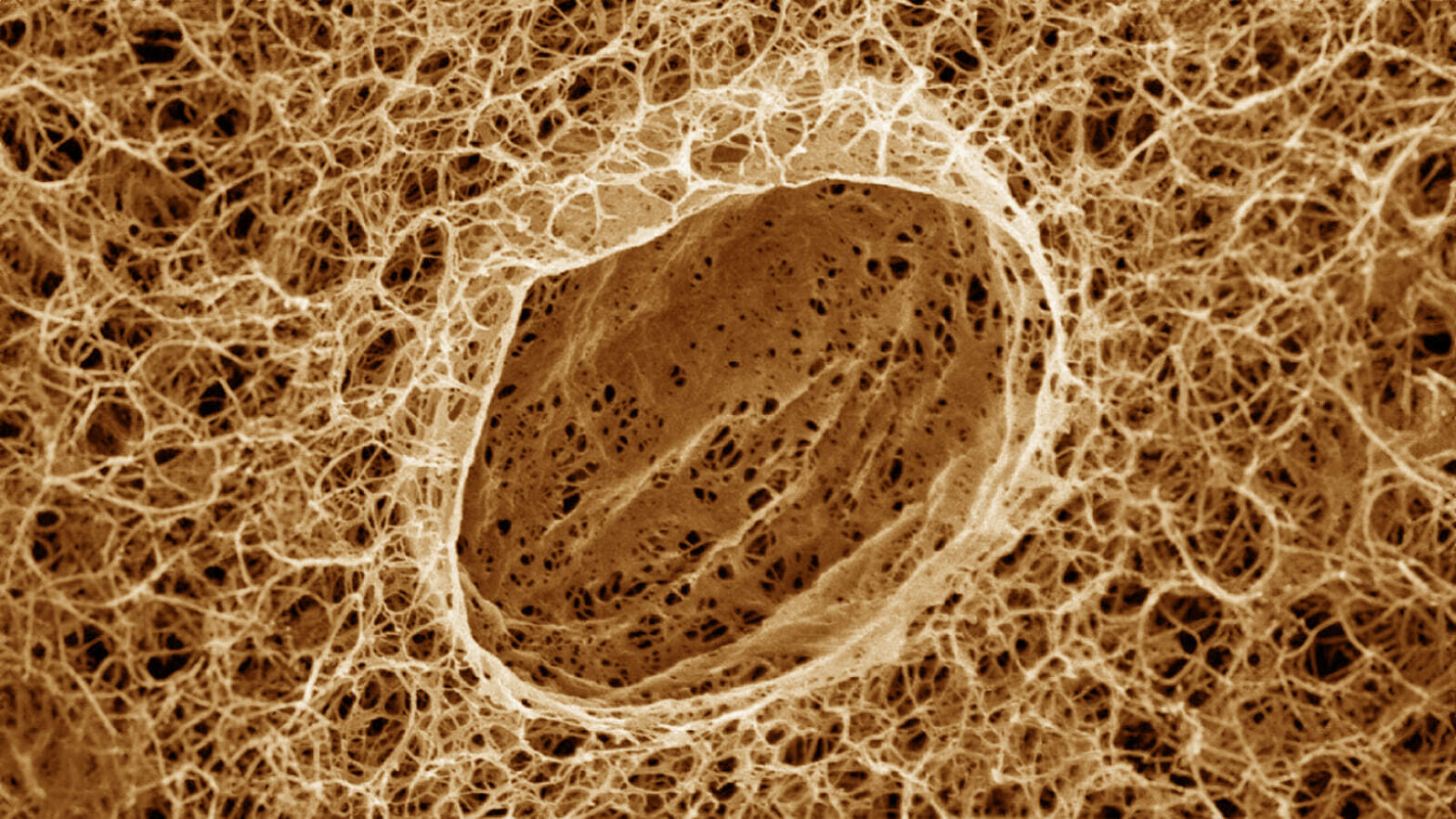
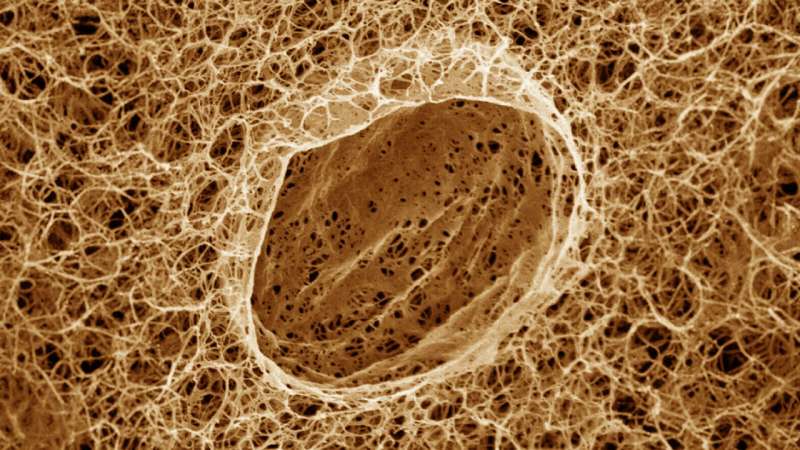
Researchers at Princeton University have demonstrated the mechanics behind a key process in living cells, where liquid droplets form and carry out complex tasks within the intricate polymer scaffolds that crisscross the cells’ interiors.
The findings provide new insight into the development of cellular condensates—organized droplets that form from cells‘ liquid content. Condensates play critical roles in health and disease, and their discovery about 15 years ago drastically changed science’s understanding of cellular operation. Many fundamental aspects of how condensates arise and grow within complex cellular environments remain poorly understood.
The study looked at how condensates’ development is affected by cells’ fibrillar networks. These networks are made of thin polymer strings that stretch throughout cells. The researchers found that condensates form in spaces between the strings in a process of liquid-liquid phase separation, akin to how oil separates from water.
The condensates apply physical pressure to the networks and then grow in big, abrupt bursts at the polymer strings fracture. Subsequent condensate growth then proceeds based on the condensates’ ability to draw in more biomolecules and the strength of the network to resist further fracturing.
This window into the nitty-gritty mechanical interactions between biomolecular condensates and fibrillar networks in cells should advance understanding on many biomedical fronts, the researchers said. In addition, the findings offer avenues for exploration in materials science toward “soft matter” substances with novel characteristics.
“Because the interiors of cells are so dense and complicated, we developed a model system to probe biomolecular condensate formation behavior in an accessible way,” said Jason Liu, a postdoctoral researcher in the lab of Rodney Priestley at Princeton and lead author of the study, published in Nature Communications in September.
Liu said the results provide insights into materials science as well as biology. Engineers are studying materials featuring liquids within fiber networks, and he said cellular behavior could help improve devices that rely on similar materials. “The insights we gained into liquid-liquid phase separation in biomolecular contexts can also be extended to new contexts, meaning we can use the ‘rules of life’ as a means toward realizing new material architectures,” Liu said.
“Jason’s demonstration of droplet formation in the presence of fibrillar networks addresses unanswered questions related to structure formation in a constrained environment that closely mimics the physical environment within a cell,” added Priestley, the Pomeroy and Betty Perry Smith Professor of Chemical and Biological Engineering at Princeton and Dean of the Graduate School.
Liu and colleagues built their cellular interior analog out of agarose hydrogels, a rubbery, gel-like substance with internal pores (spaces) akin to the pores in fibrillar networks. The researchers saturated the gels with ethanol—the alcohol present in beer, wine and liquors—and decane, a colorless, oily hydrocarbon found in gasoline.
Soaking the mixture-loaded gels in water induced phase separation of the ethanol and decane, with the decane forming biological condensate-like globs. (In real cells, condensates are composed of proteins and RNA.) The researchers included fluorescent dye, allowing the faux condensates’ growth and dynamics to be closely tracked.
Amidst this setup, small condensates arose within the agarose gel’s interconnected pore spaces, just like in a cell’s fibrillar network. The researchers observed how individual fibril-like strands in the gel confined the decane condensates’ expansion, squeezing the separating liquids into disjointed, aspherical shapes. When enough decane managed to glom onto confined condensates, strands would crack, and the condensates underwent further phase separation and swelled in size into fully formed globules.
Overall, the model demonstrated that the interplay between the fibrillar network and the phase-separating action of individual condensates ultimately helps in determining condensate quality, number, and size distribution. Getting a better intrinsic handle on how condensates naturally come about in this way will be essential for applied studies involving condensates in both living cells and non-living materials, the researchers said.
In living cells, condensate creation ties into myriad processes, from cell division to regulating gene activity and repairing DNA damage. And when condensates do not form correctly or in wrong amounts, dysfunction can ensue, studies are increasingly showing. For example, goopy and tangled condensates have been linked to neurodegenerative diseases including Alzheimer’s. Other studies have indicated likely roles for condensates in cancer, viral infections, and more. Continuing research could lead to novel therapies targeting condensate formation and function.
“Thanks to these results and additional follow-up research,” said Priestley, “we can expect improved understanding of soft matter structure formation in constrained environments, which will likely be important in many contexts of health and disease.”
From the materials science perspective, increased knowledge of liquid–liquid phase separation will inform ongoing work into solid–liquid composites. These materials feature liquid components incorporated into a matrix of fibers or other solids, and which hold promise for realizing favorable new mechanical, electrical and thermal characteristics.
“Over the long term, we can expect to learn about how to build structured matter with an unusual combination of material properties,” said Priestley.
More information:
Jason X. Liu et al, Liquid–liquid phase separation within fibrillar networks, Nature Communications (2023). DOI: 10.1038/s41467-023-41528-8
Provided by
Princeton University
Citation:
Researchers build a physical model to probe the creative destruction inside cells (2023, December 13)
retrieved 13 December 2023
from https://phys.org/news/2023-12-physical-probe-creative-destruction-cells.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.





