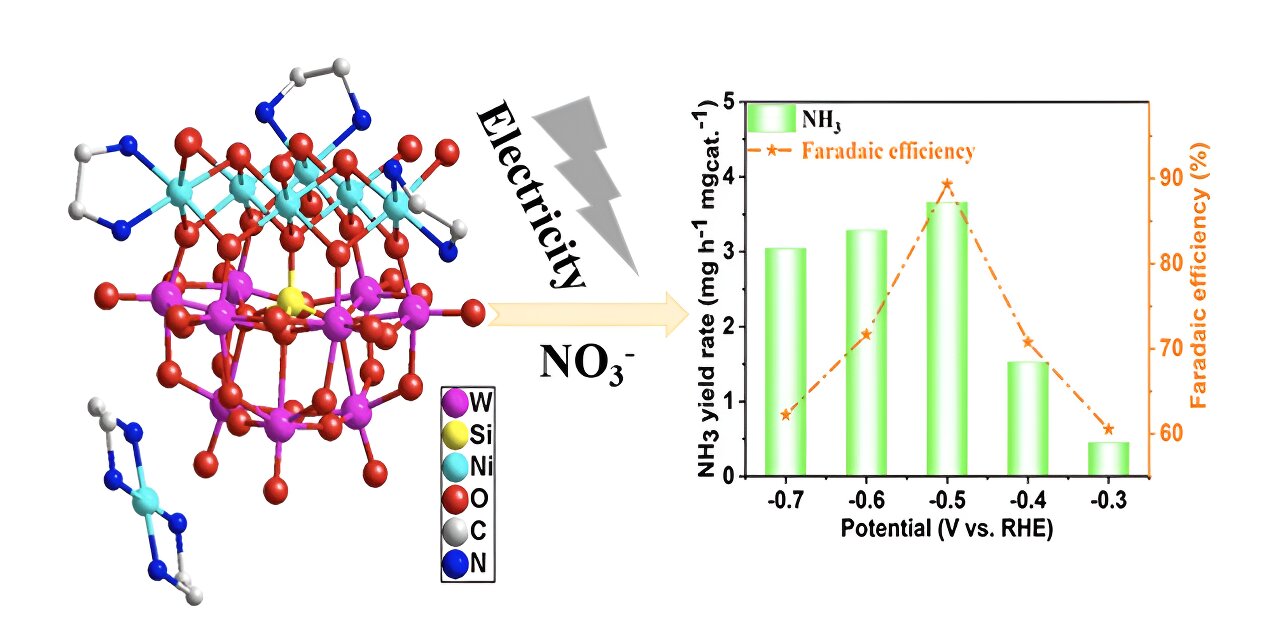
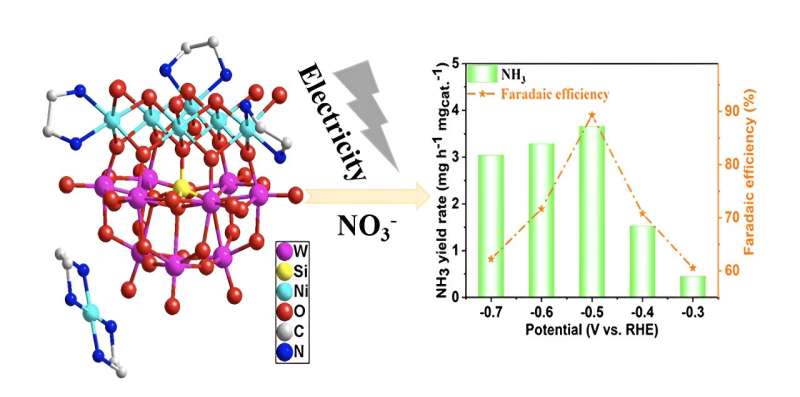
Pollution spewing from a booming global economy poses a number of different threats to human health. Researchers from Zhongyuan University of Technology proposed a new possible avenue to efficiently convert nitrate, a widespread water pollutant, back to valuable ammonia.
The team outlined a novel path to efficiently convert nitrate to ammonia using metal-added polyoxometalate as the catalyst under mild operating conditions in their study recently published in Polyoxometalates.
In the past few decades, a number of methods have been deployed to mitigate nitrate nitrogen, which contributes to groundwater contamination. Previous studies have shown that chemical reduction can not only reduce or eliminate nitrate nitrogen but can actually put the harmful pollutant to good use by converting it into ammonia—a major synthetic industrial chemical used worldwide.
Pervasive in a wide range of fields, ammonia has an extremely high energy density and is simple to liquefy and transport. A century-old method called the Haber-Bosch process converts atmospheric nitrogen to ammonia by a reaction with hydrogen using an iron metal catalyst under high temperatures and pressures. However, the pressures and temperatures necessary for the “fixation” process consume large amounts of energy and produces huge quantities of greenhouse gas emissions.
“We should find high-efficiency, environmentally friendly methods for reducing nitrogen to ammonia under mild conditions,” said Zhihui Ni, study author from the Zhongyuan University of Technology.
In the past few years, scientists have been developing a number of mild nitrogen reduction techniques as alternatives to the Haber-Bosch process, including electrocatalysis, photocatalysis, and microbial fuel cells. Of these, electrochemical nitrate reduction reaction is a promising alternative synthetic route for sustainable ammonia production, because it not only eliminates nitrate from water but also produces ammonia under mild operating conditions.
On this basis, the research team synthesized two nickel-added polyoxometalates (POMs), a class of metal-oxide clusters with unique physicochemical properties that make them particularly effective in using electrical energy to drive a chemical reaction.
Thanks to the stability of their molecular structures and reversible redox properties, POMs as catalysts can break down organic pollutants in wastewater and reduce carbon dioxide. POMs can also catalyze simple organic transformations, including bond formation.
The research team characterized the structures of the nickel compounds, and hydrothermally synthesized them to test them under high pressures. The electrochemical nitrate reduction reaction tests were performed using an electrochemical workstation.
“To evaluate the practicality of the nitrate reduction under real operating conditions, we conducted the electrocatalytic tests over a wide range of nitrate concentrations,” Ni said. They also tested for electrocatalysts’ stability, ammonia yield rate and Faraday efficiency among other parameters.
The results show high-efficiency electrochemical catalytic nitrogen reduction to ammonia.
“This discovery creates a novel path for manufacturing highly effective electrochemical nitrate reduction reaction electrocatalysts using metal-added polyoxometalate as the catalyst in ambient settings,” Ni said. “The research findings offer practical advice for creating effective electrocatalytic catalysts.”
In future steps, the research team plans to further explore this method of creating effective electrocatalytic catalysts.
More information:
Zhihui Ni et al, Hexanuclear nickel-added silicotungstates as high-efficiency electrocatalysts for nitrate reduction to ammonia, Polyoxometalates (2023). DOI: 10.26599/POM.2023.9140044
Provided by
Tsinghua University Press
Citation:
New catalyst opens efficient conversion from nitrate pollution to valuable ammonia (2023, December 14)
retrieved 14 December 2023
from https://phys.org/news/2023-12-catalyst-efficient-conversion-nitrate-pollution.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.





