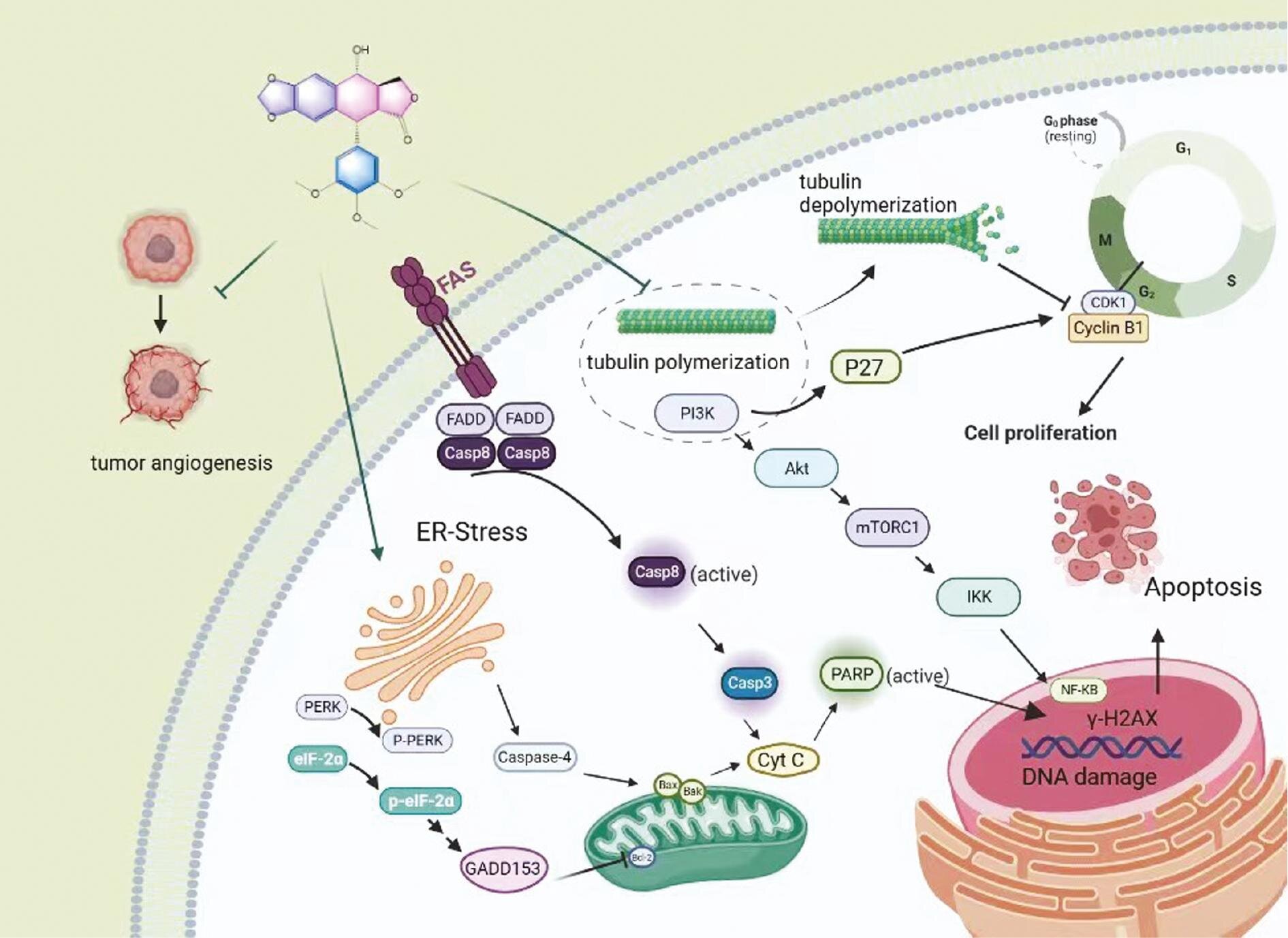
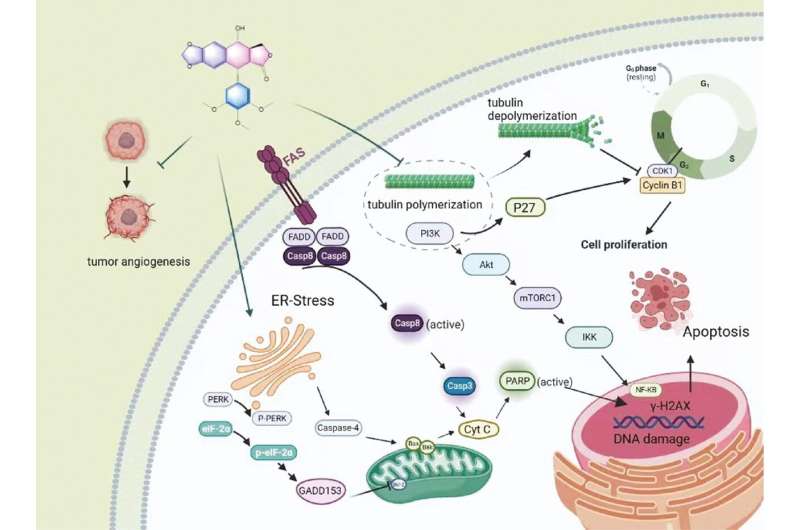
Podophyllotoxin (PPT), an aryltetralin-type lignan isolated from Podophyllum species, exhibits a wide range of biologic and pharmacologic activities, and mainly serves as an antiviral agent or antitumor drug in clinical applications.
However, the therapeutic potential of PPT has been hindered due to its detrimental systemic toxicity, poor solubility, and bioavailability. Nanoparticles, which preferentially accumulate in tumors through enhanced permeability and retention effects, have become useful tools for targeted drug delivery, thus securing a niche in cancer therapies.
The nano-based drug delivery platform has been introduced to PPT delivery for the purpose of improved solubility, enhanced efficacy, and reduced toxicity. For decades, extensive efforts have been dedicated to designing and developing various PPT delivery systems to mitigate undesirable toxicity and expand clinical applicability.
The authors of a new article published in Acta Materia Medica review the latest achievements in PPT delivery patterns and pharmacodynamic concerns with the expectation of shedding light on future research and potential applications of PPT.
More information:
Chaozheng Zhang et al, Advances in nanoscale carrier-based approaches to reduce toxicity and enhance efficacy of podophyllotoxin, Acta Materia Medica (2023). www.scienceopen.com/hosted-doc … .15212/AMM-2023-0038
Provided by
Compuscript Ltd
Citation:
Advances in nanoscale carrier-based approaches to enhance efficacy of podophyllotoxin (2023, December 13)
retrieved 13 December 2023
from https://phys.org/news/2023-12-advances-nanoscale-carrier-based-approaches-efficacy.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.





