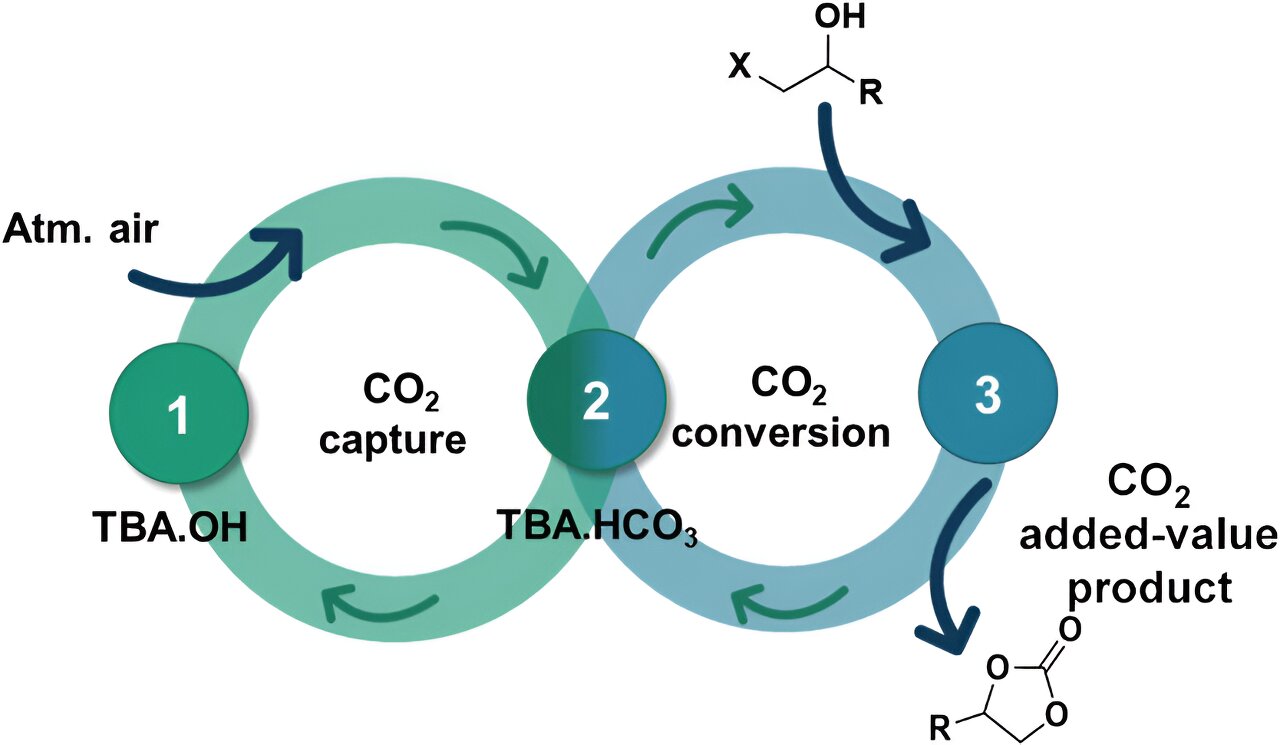
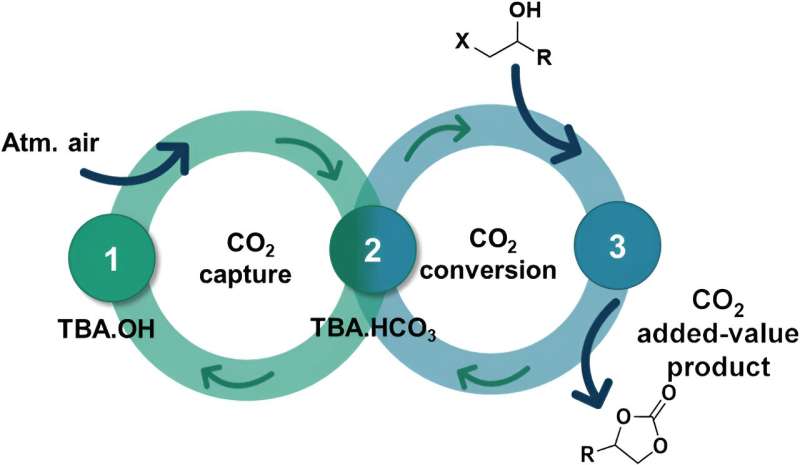
Carbon dioxide is the main contributor to greenhouse gas emissions, which are responsible for global warming and climate change. Direct capture of CO2 in the air is one of the solutions proposed to reduce its concentration in the atmosphere, but combining this process with its recovery and transformation into value-added products is highly complex and requires a finely balanced interaction between this element and the adsorbents that allow its capture and conversion into value-added products.
A research team at the Universitat Jaume I in Castelló has demonstrated a new methodology that combines the direct capture of CO2 from the air and its efficient and selective conversion into cyclic carbonates without using epoxides as substrates (which is valuable from a safety point of view as epoxides are potentially explosive).
The system uses low-cost, metal-free, commercial organic salts. The moderate reaction conditions employed (at room temperature or up to 40ºC) and the variety of substrates used demonstrate the versatility of the proposed methodology. The findings are published in the journal ACS Sustainable Chemistry & Engineering.
“The big problem and the great difficulty of this method,” explains researcher Marcileia Zanatta, “is that the CO2 in the air is at a very low concentration, approximately 0.04%, and this means that the capture reaction is very slow and its conversion into another product requires very drastic conditions.” In the system developed at the public university of Castelló, “we managed to capture it in 16 hours, which is a considerably faster rate than the approximately 40 hours reported in the literature,” states the scientist.

The development of a single material that combines catalytic and sorption capabilities to work simultaneously in both CO2 capture and conversion is a very attractive but complex challenge. In this case, a sequential process has been demonstrated, where CO2 is first captured in the bicarbonate form and then transformed into cyclic carbonates. The simplicity and low cost associated with CO2 capture, combined with the efficiency of the transformation process, open the door to a wide range of direct air capture methodologies and conversion processes (DACC).
The use of biomass-based substrates (which are also cheaper than epoxides), carbon dioxide captured directly from the air and a cheap and commercially available adsorbent “make this methodology very advantageous and attractive for developing sustainable synthetic chemical routes to generate cyclic carbonates,” says researcher Víctor Sans, “and this represents a sustainable and circular economy approach to the capture and valorization of greenhouse gases.” Sans claims that “it has been a scientific challenge to capture and transform carbon dioxide with a single system, but we have managed to create a bridge between the two.”
The production of cyclic carbonates from CO2 is very interesting from the point of view of sustainability, as it is fully efficient in atomic terms, i.e. the final product incorporates all the materials involved in the process, without creating any by-products. Cyclic carbonates are important industrial chemicals with diverse applications: environmentally friendly solvents, lithium-ion batteries, paints and coatings, resins, precursors for polymeric materials and polymer processing in fine chemistry.
More information:
Marcileia Zanatta et al, Direct Air Capture and Integrated Conversion of Carbon Dioxide into Cyclic Carbonates with Basic Organic Salts, ACS Sustainable Chemistry & Engineering (2023). DOI: 10.1021/acssuschemeng.3c00890
Provided by
Asociacion RUVID
Citation:
Researchers transform captured greenhouse gases into cyclic carbonates with biomass derivatives (2023, December 11)
retrieved 11 December 2023
from https://phys.org/news/2023-12-captured-greenhouse-gases-cyclic-carbonates.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.





