With age, cells can experience senescence, a state where they stop growing but continue releasing inflammatory and tissue-degrading molecules. When a person is young, the immune system responds and eliminates senescent cells, often referred to as zombie cells. However, zombie cells linger and contribute to various age-related health problems and diseases. Mayo Clinic researchers, in two studies, shed light on the biology of aging cells.
In a study published in Aging Cell, Mayo Clinic researchers analyzed zombie cells to explain aging at the cellular level.
“We know people age at different rates and that a person’s chronological age doesn’t always match their biological age,” says Jennifer St. Sauver, Ph.D., the lead author of the study and scientific director of the Population Health Science Scholars Program at Mayo Clinic’s Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery.
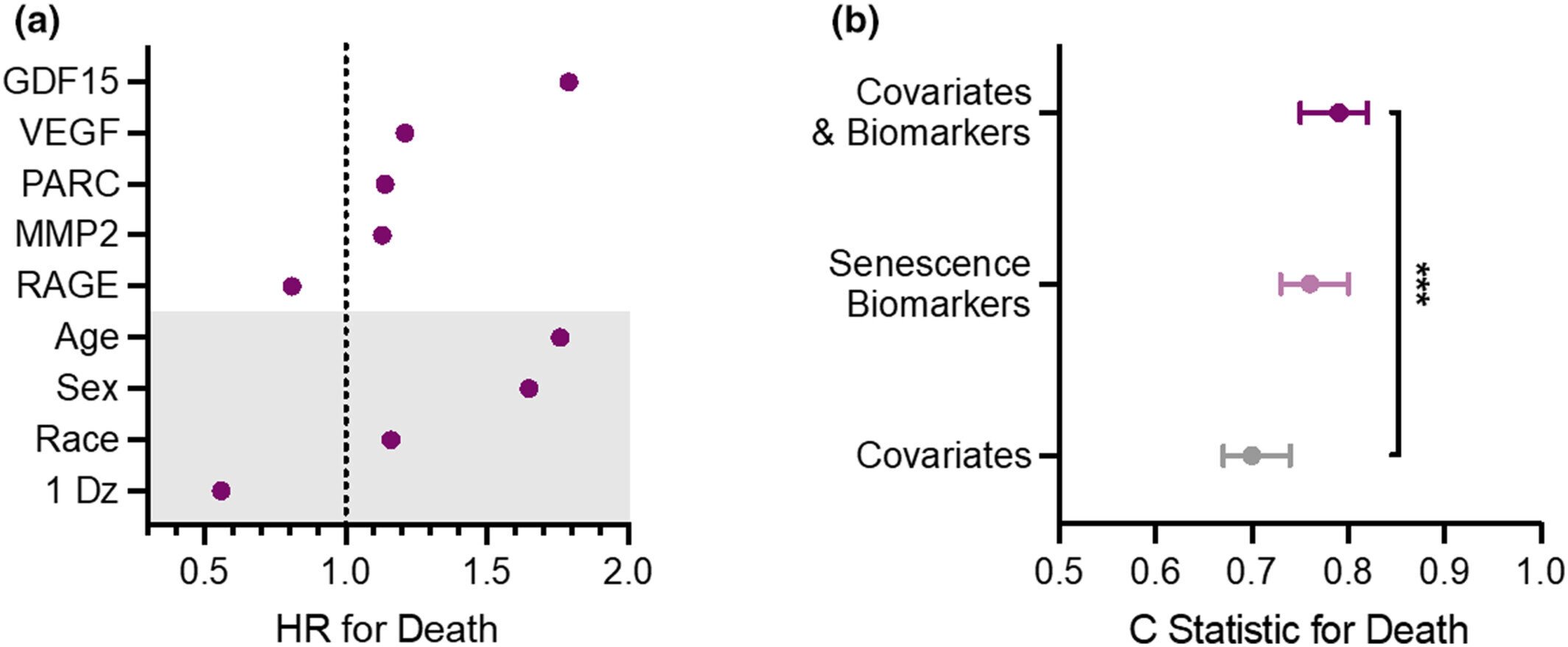
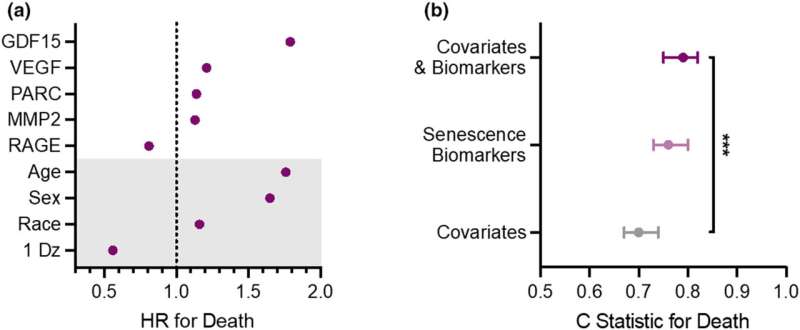
“We found that a group of diverse proteins secreted by zombie cells can serve as biomarkers of senescence and can predict health outcomes in older adults. We also found that measuring these biomarkers in the blood can help predict mortality beyond the combination of a person’s chronologic age, sex or presence of a chronic disease.”
The study included 1,923 adults aged 65 and older with one health condition or none. The group included 1,066 women and 857 men, with 68% of study participants having no chronic conditions and 32% having one condition.
“What’s so unique about this study is that even in the absence of disease, biomarkers are highly predictive of bad health outcomes in the future,” says Nathan LeBrasseur, Ph.D., director of the Robert and Arlene Kogod Center on Aging and senior author of the study. Dr. LeBrasseur studies the biological processes that drive aging.
The researchers noted that the most common chronic conditions in the group were arthritis, high cholesterol and a history of cancer.
Researchers found that higher levels of specific senescent biomarkers, such as GDF15, VEGFA, PARC and MMP2, were all associated with an increased risk of death. Some of these biomarkers have been associated with developing chronic diseases. For example, research has shown that people with heart disease and some types of cancers have higher levels of GDF15 and VEGFA. Ongoing studies are investigating how lifestyle factors, including diet, physical activity and medications that appear to help clear senescent cells, influence the circulating levels of the biomarkers.
Dr. LeBrasseur envisions using these biomarkers as tools for clinical practice to find people at risk for health challenges. In addition, research could benefit from identifying subjects who may be most responsive to emerging ways to target senescent cells and gauge their responsiveness to treatment.
Dr. St. Sauver underscores the need to include more diversity in future studies, ensuring various populations are included in ongoing aging research.
“We did see differences in levels of these biomarkers between men and women, and we also know that race and ethnicity influence many biological processes,” says Dr. St. Sauver. “For future studies, we must consider these factors in aging research.”
Uncovering unknown phenomenon in zombie cells
Mayo Clinic researcher Joao Passos, Ph.D., who also studies the biology of aging, sees his main purpose as working to enhance the vitality and health span—the period of life free from the consequences of disease and disability—in older people.
In a study published in Nature, he, along with postdoctoral researcher Stella Victorelli, Ph.D., and a large interdisciplinary team of collaborators, uncovered a previously unknown phenomenon that occurs in zombie cells.
Mitochondria, the tiny powerhouses within a cell, are responsible for producing energy but also play a crucial role when a cell incurs excessive damage. They can initiate a self-destruct mechanism called apoptosis, which leads to a cell’s death. Senescent cells, which do not die, are notorious for resisting apoptosis. These two processes, apoptosis and senescence, have often been regarded as opposite cell fates.
However, Dr. Passos, Dr. Victorelli and the team unexpectedly observed a small group of “rogue” mitochondria attempt to initiate apoptosis in senescent cells. When they do so, these mitochondria release their DNA into the cell’s cytosol, the “soup” inside a cell. Mitochondria were once independent bacteria, so the cell perceives the mitochondrial DNA as foreign, which sparks inflammation that can damage tissues and lead to disease.
Furthermore, the researchers found that if they blocked this process in mice equivalent in age to a 70-year-old human, they could reduce tissue inflammation and significantly boost their health, including improving their strength, balance and bone structure.
This newfound knowledge of the inner workings of a senescent cell may open doors to new types of therapeutics, beyond senolytics that eliminate senescent cells, to improve peoples’ health span.
“Inflammation is one of the main ways we think senescent cells cause detrimental effects in aging-related diseases. So, it’s exciting to have found a way to target a pathway that reduces inflammation,” Dr. Victorelli explains.
Mapping out the big picture
Dr. Passos and colleagues, in collaboration with researchers across the U.S., are part of an ambitious project to map senescent cells across different tissues. The project is called the Cellular Senescence Network (SenNet). Its primary objective is to compile a comprehensive atlas of senescent cells within the human body over a lifetime. Dr. Passos recently spearheaded an effort published in Nature Aging to highlight emerging technologies and challenges in spatial mapping of cellular senescence.
“By targeting these fundamental mechanisms driving the aging process rather than tackling each disease individually, we have the potential to not only fight one disease of aging but all of them,” Dr. Passos says.
More information:
Jennifer L. St. Sauver et al, Biomarkers of cellular senescence and risk of death in humans, Aging Cell (2023). DOI: 10.1111/acel.14006
Stella Victorelli et al, Apoptotic stress causes mtDNA release during senescence and drives the SASP, Nature (2023). DOI: 10.1038/s41586-023-06621-4
Citation:
Shedding light on health and zombie cells in aging (2023, December 4)
retrieved 4 December 2023
from https://phys.org/news/2023-12-health-zombie-cells-aging.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.





