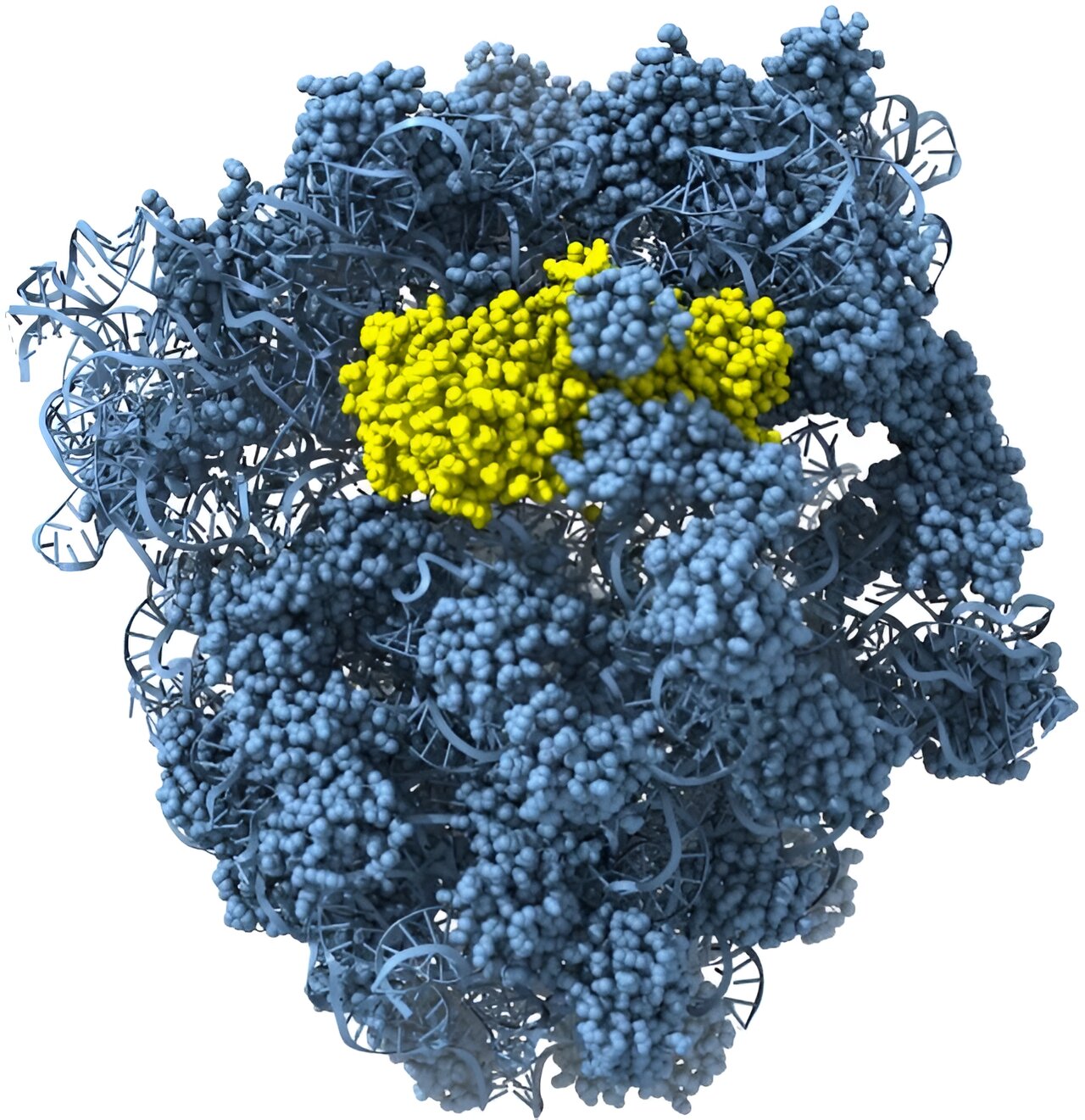
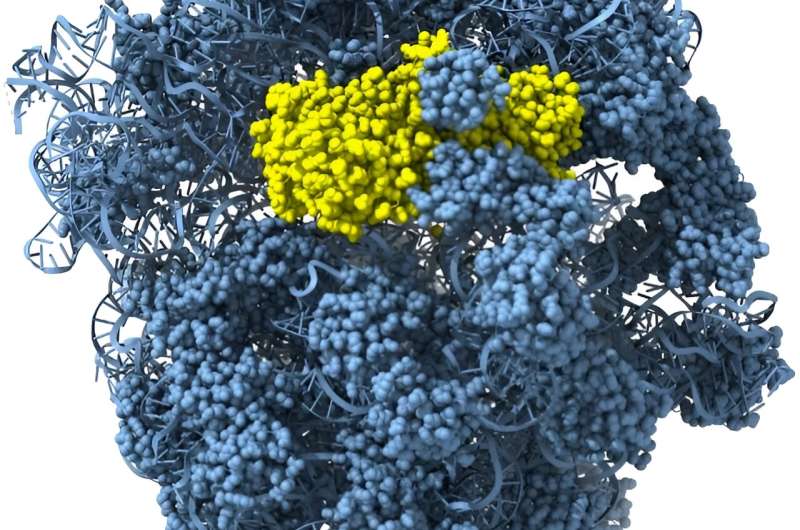
A recent study in Science uses bioinformatic analysis as well as biochemical and structural studies to characterize a novel family of effector proteins, named Cami1. The research shows that when a virus attacks a bacterium, CRISPR-Cas10 signaling molecules activate Cami1—a ribosome-dependent ribonuclease.
“Activated Cami1 cleaves mRNAs that are involved in protein synthesis, thereby inhibiting cell growth. This allows the bacterium to save resources and prevents production of the viral proteins,” says Gintautas Tamulaitis, who led the the Vilnius University researchers.
Using X-ray structural analysis and cryo-electron microscopy (cryo-EM), researchers determined structures of both apo-Cami1 and the Cami1 complex with the protein synthesis machine—the ribosome. Structural studies provided insights into how Cami1 can specifically cleave mRNA.
It was shown that Cami1’s interaction with a specialized ribosome structure, called the ribosomal stalk, is necessary for its entry into the protein synthesis center.
“Interestingly, the same capture mechanism to bind the ribosome is used by plant antiviral proteins that also inactivate ribosomes. This discovery unveiled an additional layer of the CRISPR-Cas antiviral defense system and demonstrated a common antiviral strategy shared between eukaryotes and bacteria. Knowledge about our characterized Cami1 proteins will contribute to the development of new molecular tools in biotechnology and therapy,” says Gintautas Tamulaitis.
The ribosome-Cami1 complex was visualized using a 200 kV Glacios Cryo-Transmission Electron Microscope, acquired by Vilnius University in 2020. The first findings from this microscope were published in Nature earlier in 2023.
More information:
Irmantas Mogila et al, Ribosomal stalk-captured CARF-RelE ribonuclease inhibits translation following CRISPR signaling, Science (2023). DOI: 10.1126/science.adj2107
Provided by
Vilnius University
Citation:
Researchers characterize translation inhibition in CRISPR-Cas antiviral defense system (2023, December 4)
retrieved 4 December 2023
from https://phys.org/news/2023-12-characterize-inhibition-crispr-cas-antiviral-defense.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.





